Remethylation Disorders Therapeutic Avenues
The current therapeutic objective is described in the Guidelines for the diagnosis and management of Remethylation Disorders. Click here to view the Guidelines.
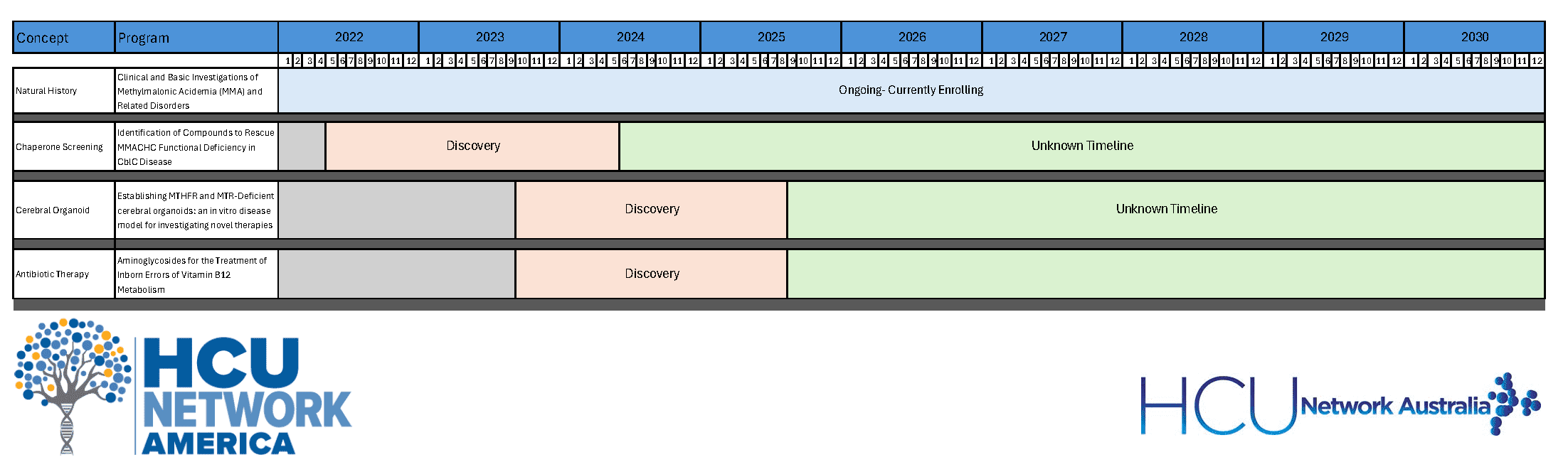
There are currently three HCUNA-funded projects for the treatment of Remethylation disorders being investigated. The majority of current research is focused on the natural history and improving diagnosis and treatment. The following are descriptions of potential avenues of research that have promise for novel therapeutics.
NATURAL HISTORY STUDIES
Natural history studies are imperative in understanding the biochemical pathway for Remethylation disorders to aid in diagnosis and treatment. Following patients for an extended period of time, researchers analyze treatment protocols, clinical manifestations, and medication management to improve patients' quality of life. Here are two natural history studies that will help further the understanding of Remethylation Disorders.
- Clinical and Basic Investigations of Methylmalonic Acidemia and Related Disorders.
- Currently enrolling patients over age 2, the National Institute of Health is conducting a natural history study of remethylation disorders. For more information click here.
- Longitudinal study of ultra-rare inherited metabolic and degenerative neurological diseases.
- Currently enrolling patients over 18 who live in Europe, this study has a larger focus area but includes remethylation disorders due to their ultra-rare status and degenerative neurological issues associated with the disorder.
Potential Treatment Pathways
Experts take different perspectives as to which type of new treatment modalities should be pursued with higher priority. Due to the different defects that cause homocystinuria, one approach will likely not create a “cure-all.” Rather, understanding and improving our understanding of the basic science of the pathway will lead to a greater understanding and the possibility of finding a novel approach to improve clinical outcomes. The following areas of future research are options to explore future treatment options.
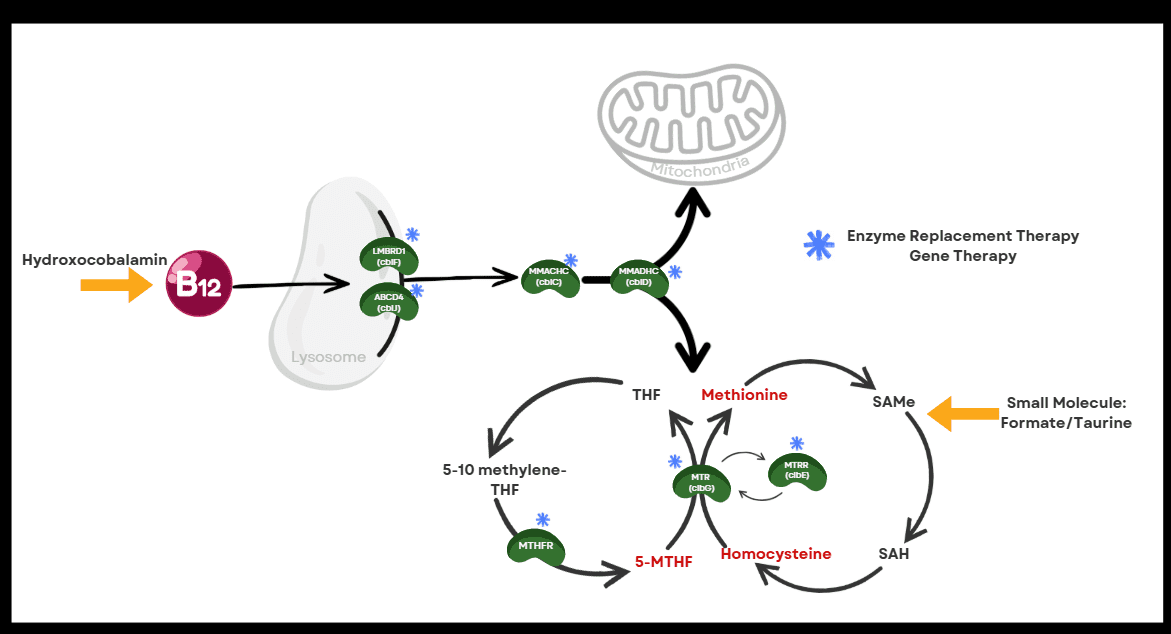
Here is a schematic overview showing the biochemical pathway and how current therapeutic interventions address the biochemical or disease process.